Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons. Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons. You can effortlessly find every single detail about the elements from this single Interactive Periodic table. Another common method of categorization recognizes nine element families: Alkali Metals: Group 1 (IA) - 1 valence electron. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Free Gift for you: Interactive Periodic Table The first electron affinity is the energy released when 1 mole of gaseous atoms each acquire an electron to form 1 mole of gaseous -1 ions. In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. Electron affinities are the negative ion equivalent, and their use is almost always confined to elements in groups 16 and 17 of the Periodic Table. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell).

The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. There are total 18 vertical columns on periodic table. This is the reason why H is always a terminal atom and never a central atom.Groups are the vertical columns on the periodic table. Hydrogen has only one valence electron and can form only one bond with an atom that has an incomplete outer shell. Group Valence 1 Valence 2 Valence 3 Valence 4 Valence 5 Valence 6 Valence 7 Valence 8 Typical valences 1 (I) NaCl KCl: 1. Hydrogen only needs to form one bond to complete a duet of electrons. For elements in the main groups of the periodic table, the valence can vary between 1 and 8. The elements in a group have similar physical or chemical characteristics of the outermost electron. The largest atomic radius of elements in their period. There are 18 numbered groups in the periodic table the 14 f-block columns, between groups 2 and 3, are not numbered. Highly reactive, with reactivity increasing moving down the group. Atom (Group number)īecause hydrogen only needs two electrons to fill its valence shell, it follows the duet rule. In chemistry, a group (also known as a family) 1 is a column of elements in the periodic table of the chemical elements. Table showing 4 different atoms, each of their number of bonds, and each of their number of lone pairs. In each case, the sum of the number of bonds and the number of lone pairs is 4, which is equivalent to eight (octet) electrons. Each new period begins with one valence electron. The rule is as follows: If an element is not a transition metal, then valence electrons increase in number as you count groups left to right, along a period. The number of electrons required to obtain an octet determines the number of covalent bonds an atom can form. Apply the rule of the periodic table to your element. Oxygen and other atoms in group 16 obtain an octet by forming two covalent bonds: To obtain an octet, these atoms form three covalent bonds, as in NH 3 (ammonia). Group 15 elements such as nitrogen have five valence electrons in the atomic Lewis symbol: one lone pair and three unpaired electrons. The transition elements and inner transition elements also do not follow the octet rule since they have d and f electrons involved in their valence shells. Atoms share electrons and form covalent bonds to satisfy the octet rule. 4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. The atoms in group 6A make two covalent bonds.
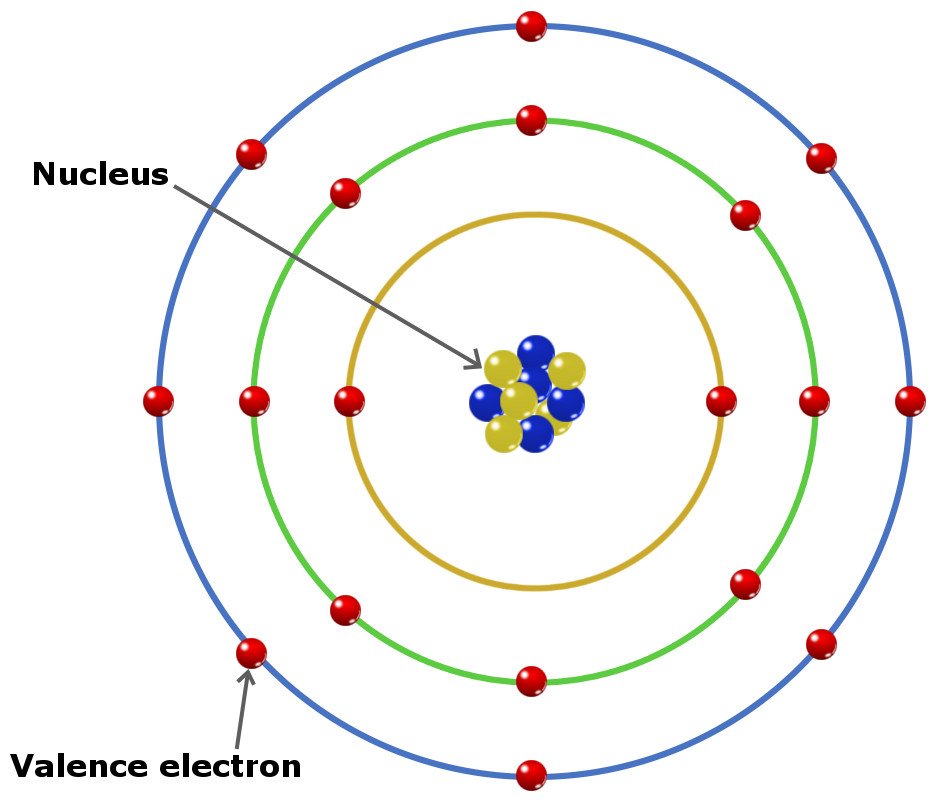
Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule and only needs to form one bond. Covalent bonds are formed by two atoms sharing electrons.
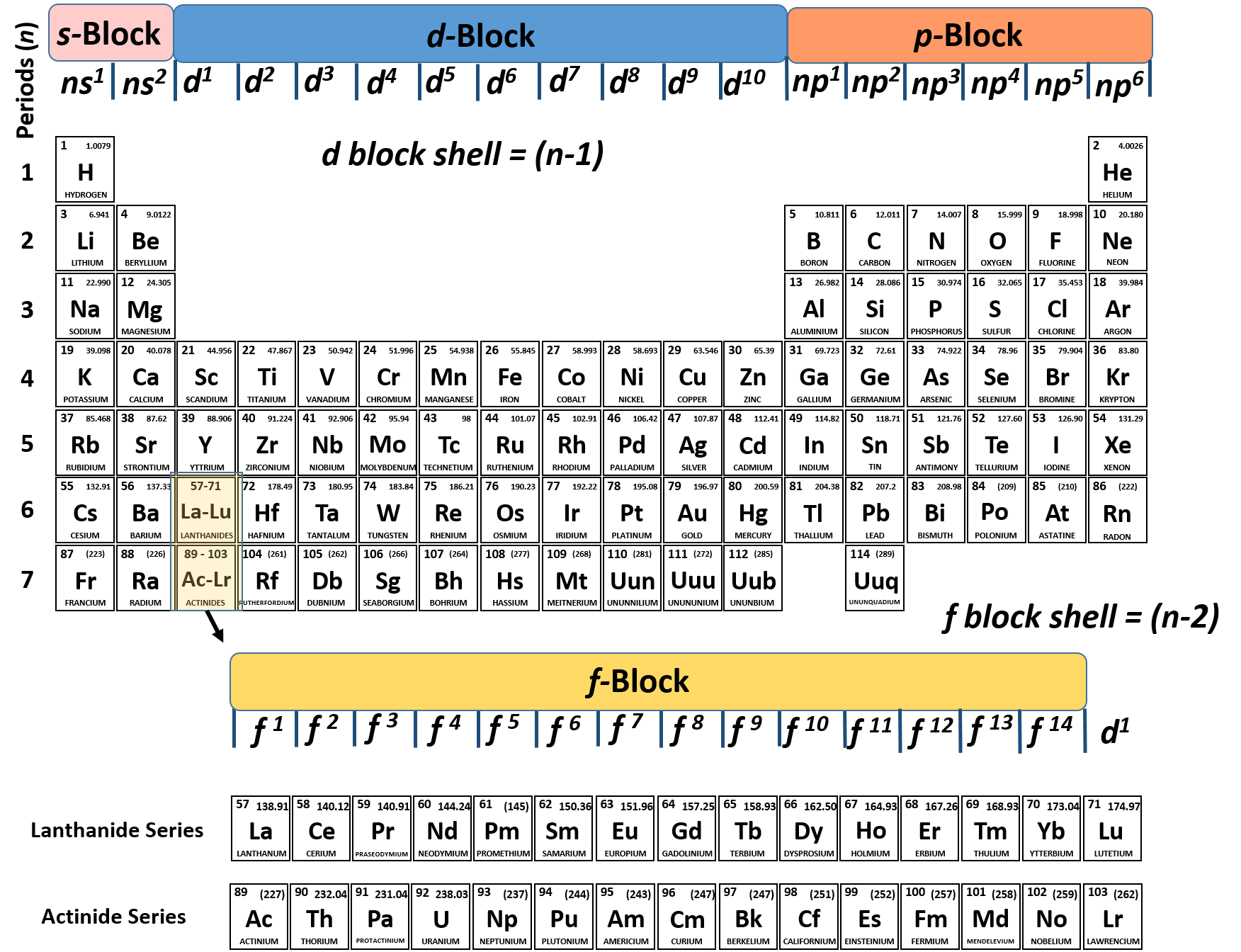
Answer: All have an ns2np5 electron configuration, one electron short of a noble gas electron configuration. Use the periodic table to predict the characteristic valence electron configuration of the halogens in group 17. These four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in CCl 4 (carbon tetrachloride) and silicon in SiH 4 (silane). By extrapolation, we expect all the group 2 elements to have an ns2 electron configuration. For example, each atom of a group 14 element has four electrons in its outermost shell and therefore requires four more electrons to reach an octet. The trend for electron affinity values across periods and down groups is shown in this version of the periodic table. The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). From left to right: water molecule, ammonia molecule, and methane molecule
